"B cell and autoimmunity" research group
„B cell and autoimmunity” research group
Head of the research group
Gabriella Sármay, Ph.D., D.Sc., Professor (retired), professor emerita
Members of the research group
Gyebrovszki Balázs, PhD student
Mustafa Talib, SH PhD student
Bőgér Dorottya, 2nd year Msc student
Research interest
The focus of my interest is the regulation of the B cell immune response, the intracellular signaling triggered by B cell receptors and the "cross-talk" between activatory and inhibitory receptors, as well as the pathomechanism of autoimmune disease, such as rheumatoid arthritis. The signals mediating negative selection of B cells play a role in maintaining immunological tolerance. Defective regulation can lead to the breakdown of tolerance and eventually, the development of autoimmune diseases.
In our previous research, we investigated the function of Fcγ receptors (FcγR) that recognize the constant (Fc) part of IgG, we identified the kinases and phosphatases that are responsible for the development of B-cell inhibition mediated by FcγRIIb, furthermore we studied the differences between the signal transduction processes in immature and mature B cells, as well as the role of the adapter protein Grb2-associated binder, a key player in the regulation of signal transduction.
In recent years, our attention has turned to a systemic autoimmune disease, Rheumatoid arthritis (RA). RA is an inflammatory autoimmune disease associated with the degradation of cartilage and bone in small joints, and currently cannot be cured. Antibodies against post-translationally modified, citrullinated proteins (anti-citrullinated protein/peptide antibodies, ACPA) are present in the serum of approximately 70% of the patients, detection of ACPA in sera of patients is the most sensitive diagnostic method of RA. Additionally, ACPA has a pathological significance in the disease.
Research projects:
THE MULTIPLE LEVEL OF IMMUNOREGULATION IN RHEUMATOID ARTHRITIS: THE ROLE OF B CELLS
In our previous project entitled: THE MULTIPLE LEVEL OF IMMUNOREGULATION IN RHEUMATOID ARTHRITIS: THE ROLE OF B CELLS (NKFIH, NK 104846, K 104385)
1.) We designed and synthesized bispecific multivalent peptide constructs that were recognized by BCR on one hand and triggered effector mechanisms on the other. These constructs were coupled to biodegradable nanoparticles and successfully used in ex vivo tests to eradicate ACPA producing B cells. Such constructs would allow the development of potential therapies designed to eliminate B-cells responsible for ACPA production.
- Recognition of new citrulline-containing peptide epitopes by autoantibodies produced in vivo and in vitro by B cells of rheumatoid arthritis patients. Szarka E, Babos F, Magyar A, Huber K, Szittner Z, Papp K, Prechl J, Pozsgay J, Neer Z, Ádori M, Nagy G, Rojkovich B, Gáti T, Kelemen J, Baka Z, Brózik M, Pazár B, Poór G, Hudecz F, Sármay G. Immunology. 2014.141(2):181-91.
- In vitro eradication of citrullinated protein specific B-lymphocytes of rheumatoid arthritis patients by targeted bifunctional nanoparticles. Pozsgay J, Babos F, Uray K, Magyar A, Gyulai G, Kiss É, Nagy G, Rojkovich B, Hudecz F, Sármay G. Arthritis Res Ther. 2016. 16;18:15
- Affinity Purification and Comparative Biosensor Analysis of Citrulline-Peptide-Specific Antibodies in Rheumatoid Arthritis. Szarka E, Aradi P, Huber K, Pozsgay J, Végh L, Magyar A, Gyulai G, Nagy G, Rojkovich B, Kiss É, Hudecz F, Sármay G. Int J Mol Sci. 2018. 19(1):326.
- Biologia Futura: Emerging antigen-specific therapies for autoimmune diseases. Sármay G. Biol Futur. 2021. 72(1):15-24.
2.) To reveal the mechanism of B-cell regulation by FcɣRIIb, in collaboration with Xencor Inc. (Monrovia, Ca, US) Fc-engineered antibodies (FcγRIIbE) with enhanced binding capacity to FɣRIIb were tested, and the consequences on B-cell activation were monitored. FcγRIIbE anti-CD19 Xmab5871 antibody inhibited BCR and/or CD19 mediated signaling without killing the cells. The antibody also inhibited differentiation of citrullinated peptide-specific plasma cells from rheumatoid arthritis patients. We gained new information on the interaction between CD19, FcγR and BCR, and on the mechanism of B-cell inhibition by FcγRE antibodies, possibly applicable for RA therapy.
- Suppression of innate and adaptive B cell activation pathways by antibody coengagement of FcγRIIb and CD19. Szili D, Cserhalmi M, Bankó Z, Nagy G, Szymkowski DE, Sármay G. MAbs. 2014.6(4):991-9.
3.) In another work we studied the role of TGFβ activated kinase (TAK1) in BCR and TLR9 signaling and concluded that TAK1 is a key regulator of receptor crosstalk between BCR and TLR9 thus plays a critical role in B cell development and activation.
- TGFβ activated kinase 1 (TAK1) at the crossroad of B cell receptor and Toll-like receptor 9 signaling pathways in human B cells. Szili D, Bankó Z, Tóth EA, Nagy G, Rojkovich B, Gáti T, Simon M, Hérincs Z, Sármay G. PLoS One. 2014. 9(5): e96381.
4.) We have compared IL-10 producing regulatory B cells (B10 cells) from healthy persons and RA patients and found that the TLR9 and CD40 mediated combined stimuli were highly efficient at inducing human B10 cells, which primarily originate from the CD19+ CD27+ subset. We found a significantly lower number of B10 cells in the CpG+CD40L-stimulated samples from RA patients that had a reduced capacity to inhibit IFNɣ production by T cells. On the other hand, we did not detect a significant difference between B10 cells in RA patients and healthy subjects when IL-21 was also added. We also found that TLR9- and CD40-activated CREB and IL-21–activated STAT3 play a synergistic role in signaling pathways that induce B10 cells. IL-21 expanded the B10 population and induced the differentiation of CD27+ B10 cells into Blimp-1+ regulatory plasmablasts.
- Regulatory B cells in rheumatoid arthritis: Alterations in patients receiving anti-TNF therapy. Bankó Z, Pozsgay J, Gáti T, Rojkovich B, Ujfalussy I, Sármay G. Clin Immunol. 2017.184:63-69
- Antigen-specific immunotherapies in rheumatic diseases. Pozsgay J, Szekanecz Z, Sármay G. Nat Rev Rheumatol. 2017. 13(9):525-537.
- Induction and Differentiation of IL-10-Producing Regulatory B Cells from Healthy Blood Donors and Rheumatoid Arthritis Patients. Bankó Z, Pozsgay J, Szili D, Tóth M, Gáti T, Nagy G, Rojkovich B, Sármay G. J Immunol. 2017. 198(4):1512-1520.
- MZ B cells migrate in a T-bet dependent manner and might contribute to the remission of collagen-induced arthritis by the secretion of IL-10. Huber K, Sármay G, Kövesdi D.Eur J Immunol. 2016. 46(9):2239-46.
- Inflammatory signal induced IL-10 production of marginal zone B-cells depends on CREB. Barátki BL, Huber K, Sármay G, Matkó J, Kövesdi D. Immunol Lett. 2019. 212:14-21.
COMPARATIVE STUDY OF PROTEIN GLYCOSYRATION IN RHEUMATOID ARTHRITIS
Another NKFIH-supported project (K16, 119549, COMPARATIVE STUDY OF PROTEIN GLYCOSYRATION IN RHEUMATOID ARTHRITIS) in collaboration with MTA TTK (Prof. Dr. Károly Vékey) we investigated the glycosylation patterns of ACPA and IgG from healthy individuals and studied the differences in the proinflammatory functions of various IgG glycoforms. We found that both sialylation and galactosylation levels of ACPA-IgG negatively correlate with inflammation-related parameters such as CRP, ESR, and RF. Functional assays show that dimerized ACPA-IgG significantly enhances TNFα release from U937 cells in an FcγRI-dependent manner, whereas healthy IgG does not. TNFα production inversely correlates with the relative intensities of the G0 glycoform, which lacks galactose and terminal sialic acid moieties.
- The Role of IgG Fc Region N-Glycosylation in the Pathomechanism of Rheumatoid Arthritis.Gyebrovszki B, Ács A, Szabó D, Auer F, Novozánszki S, Rojkovich B, Magyar A, Hudecz F, Vékey K, Drahos L, Sármay G. Int J Mol Sci. 2022 May 23;23(10):5828.
COOPERATION BETWEEN REGULATORY B CELLS AND FOLLICULAR T CELL SUBSETS IN HEALTHY SUBJECTS AND IN INFLAMMATORY AUTOIMMUNE DISEASE
In the present on-going NKFIH supported project (K18, 128546): COOPERATION BETWEEN REGULATORY B CELLS AND FOLLICULAR T CELL SUBSETS IN HEALTHY SUBJECTS AND IN INFLAMMATORY AUTOIMMUNE DISEASE, our hypothesis is that the defective interactions between T and B cells may contribute to the development of RA. The strictly regulated function of the immune system can sometimes fail, and the immune cells may break tolerance and turn against their own tissues, inducing inflammation and tissue damage. Our aim is to find out whether there is any difference between RA patients and healthy people in terms of the cooperation between the individual regulatory B- and T-cells (Breg, Tfr and Tfh). We would like to explore the faulty regulatory mechanisms, and to see if there is a correlation between the possible deviations and the severity and course of the disease. Our expected results will contribute to a better understanding of the pathomechanism of RA and the development of more specific diagnostics/therapy in the future.
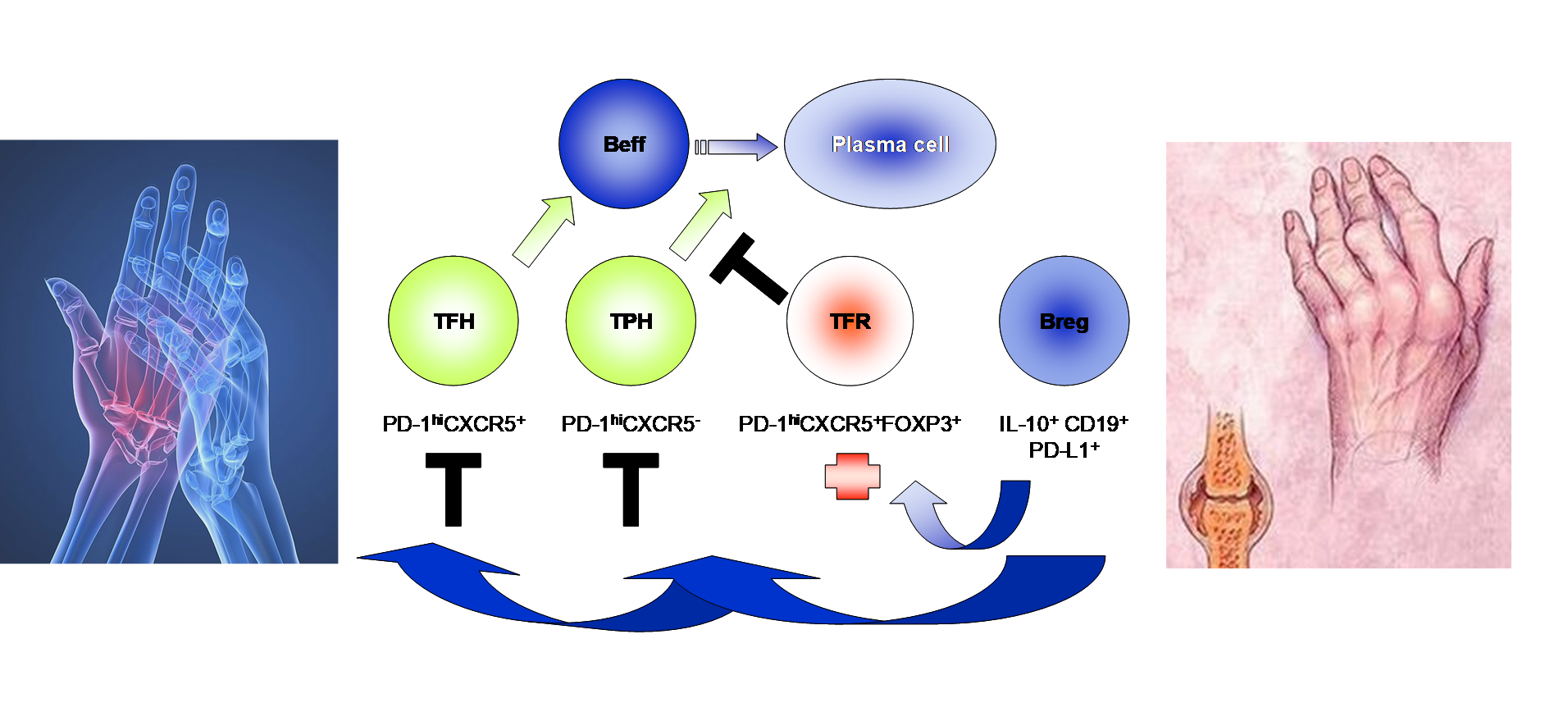
Hypothesis: Defective interactions between T and B cells may contribute to the development of RA.
Beff: effector B-cell, TFH: follicular helper T-cell, TPH: peripheral helper T-cell, TFR: follicular regulatory T-cell, Breg: regulatory B-cell

